 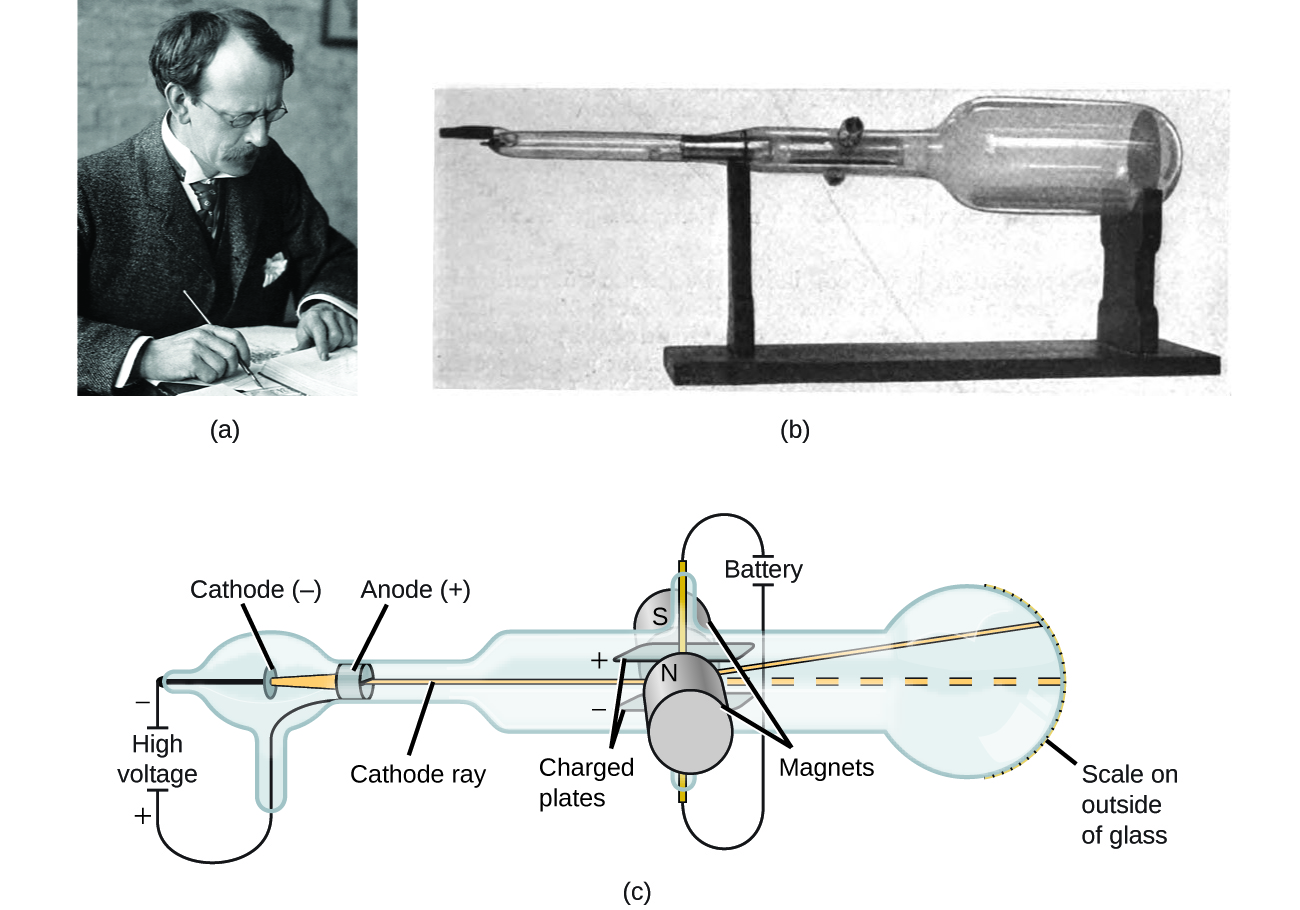
The charge-to-mass ratio could then be calculated, and was found to be at least 1,000 times higher than that of the hydrogen atom. An equal deflection was produced by applying a magnetic field, perpendicular to the electric field, using an electromagnet. Their work culminated in the discovery by English physicist J.J. The beam was deflected by passing it through an electric field between the parallel plates. In the apparatus shown here, rays produced at the cathode were focused into a narrow beam by the slits in the cylinders in the narrow part of the tube, producing a fluorescent spot on the spherical end of the tube. Thomson‘s historic Royal Institution lecture of April 30, 1897, on Cathode Rays, has been reproduced in full in the November-December issue of the American Journal of Physics as the contribution of the American Association of Physics Teachers to the Electron Jubilee celebrations (see also Nature, 160, 776 1947). By 1897, Thomson had measured the charge-to-mass ratio, but finding the charge or mass of the electron independently took a further two years. Others, such as geophysicist Emil Wiechert, realized that the charged carriers of electricity were very small, but Thomson seems to have been the first to suggest that electrons - or 'corpuscles', as he called them - existed inside the atom he put forward a model for atomic structure that was based on the patterns of repulsion of floating magnets noted by Alfred Mayer in 1878. This earlier generation had observed that these rays travelled in straight lines but could be deflected by a magnetic field, which became the basis of Thomson's work. Thomsons positive ray apparatus were analogous to his cathode-ray tubes (see objects EM.N-08013A and EM. He was investigating a long-standing puzzle known as. At the Cavendish Laboratory at Cambridge University, Thomson was experimenting with currents of electricity inside empty glass tubes. Thomson was venturing into the interior of the atom. Thomson (18561940) proved that atoms were not the most basic form of matter. Image used with Permission (CC BY-SA-NC).   
The discharge tubes and pumps invented by Heinrich Geissler in the 1850s, and the induction coil perfected by Heinrich Ruhmkorff at about the same time, had enabled many physicists, such as Julius Plücker and Johann Hittorf, to experiment with the new 'cathode rays'. 17), British physicist, president of the Royal Society, teacher of Ernest Rutherford and winner of the Nobel Prize in physics in the year 1906, succeeded in deciphering the nature of the cathode rays by conducting different experiments. ne hundred years ago, amidst glowing glass tubes and the hum of electricity, the British physicist J.J. When a high voltage is applied to a gas contained at low pressure in a gas discharge tube, electricity flows through the gas, and energy is emitted in the form of light. The road leading to the discovery had been an arduous one, trodden by many scientists. Although it was certainly one of his instruments, he used a series of cathode-ray tubes in that year, so we cannot be certain.Īlthough the moment of discovery of the first subatomic particle has been controversial, Thomson's measurement of the charge-to-mass ratio for cathode rays is now considered to have defined the electron. This has been claimed to be the apparatus with which Joseph John Thomson discovered the electron in 1897. Thomson - the Centenary of His Discovery of the Electron and his invention of Mass Spectrometry, Rapid Communications in Mass Spectrometry, Vol.11, 2-16 (1997).Round and round, faster and faster. Thomson, the Cavendish Laboratory, and Thomson's cathode ray tube and positive ray apparatus, see J. With it, the traces were recorded photographically for the first time. This object is a replica of the fourth such apparatus Thomson constructed. This beam of "positive rays" is then deflected vertically by a magnetic field and horizontally by an electric field, forming a parabolic trace on a phosphorescent screen-all atoms or molecules of the same charge-to-mass ratio falling along one parabola. Ions, created by an electric discharge in the glass bulb, are accelerated towards its mouth. Thomson's positive ray apparatus were analogous to his cathode-ray tubes (see objects EM.N-08013A and EM.N-08013B) in that they measured the ratio of charge to mass of "positive rays" instead of electrons (cathode rays). Thomsons experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. The rays fall on a phosphorescent screen at the other end of the apparatus, where a photographic plate can also be positioned. The chamber contains a pair of plates for deflection of the beam of "positive rays" (ions) passing through from the discharge tube. Wood base supports, at one end, a horizontal glass discharge tube attached to a chamber fitted between the poles of an electromagnet. Object is a replica of the original which resides at Cavendish Laboratory, Cambridge University, England. Object ID EM.N-08014 Overall length 62 cm x width 35 x height 44 cm. Thomson's positive ray apparatus, replica of Cavendish Lab apparatus. Thomson's positive ray apparatus (B&W) Previous Next Description: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
